
What is SPRAVATO®?
SPRAVATO® is the only FDA-approved nasal spray for Treatment Resistant Depression (TRD). SPRAVATO® is self-administered under the supervision of a healthcare professional.
What to Expect
How to Prepare
- Do not eat at least 2 hours before taking SPRAVATO®, and do not drink liquids at least 30 minutes before taking SPRAVATO®.
- Corticosteroids and nasal decongestant medications should be taken no later than 1 hour before taking SPRAVATO®.
Treatment Experience
- Patient will check-in with the provider during each appointment and the provider will remain on-site for the duration of treatment.
- Each appointment is approximately 2.5 hours in length.
- There is a minimum observation period of 2 hours after the medication has been self-administered.
- Treatments are facilitated in a shared but private, low-stimulus environment with reclining chairs.
- Patients are advised to stay seated during treatment for safety and stability purposes.
- We recommend bringing personal items, such as a phone, tablet, music player, headphones, crossword puzzle or book to pass time.

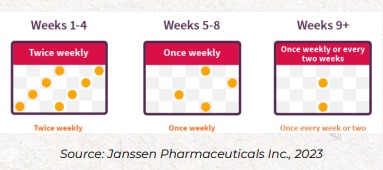
How Long Do I Have to Take SPRAVATO®?
The first phase of treatment is known as the “Induction Phase,” where the patient treats twice weekly for the first 4 weeks. After the induction phase, after reassessment with the overseeing psychiatric provider, the patient either continues treatment on a “maintenance” basis (typically treating once weekly or every other week), or is discharged from treatment (if the patient has not received any response or benefit from the treatment during the Induction Phase).
Most Common Side Effects
Most common side effects of SPRAVATO® include the following: dissociation, dizziness, numbness, sedation, spinning sensation, decreased feeling of sensitivity, feeling anxious, lack of energy, increased blood pressure, vomiting, feeling drunk, or feeling very happy or excited.
If these common side effects occur, they usually happen right after taking SPRAVATO® and go away the same day.
These are not all the possible side effects of SPRAVATO® Source: https://www.spravato.com/taking-spravato
After Your Treatment
After your SPRAVATO® treatments, patients are not be able to drive, operate machinery or do anything that requires being “fully alert”, so transportation home from treatment must be established prior to your SPRAVATO® appointment. Potential side affects may affect mental alertness and motor coordination, so patients are advised to wait until the next day and following restful sleep before resuming any normal day-to-day activities. Good options include a friend or family member, utilizing a Ride Share program such as Uber or Lyft, or public transportation.



